Nature Chemistry
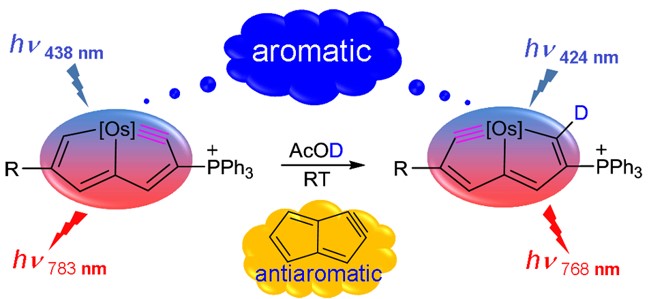


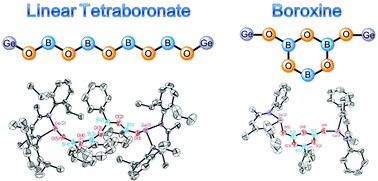
Two newly discovered linear compounds tetraboronate and boroxine stabilized by digermylene are reported, which feature a B4O5 chain and a B3O3 ring, respectively. DFT calculations reveal that not only can digermylene stabilize the electron-deficient boron centers, but also increase the energies of the LUMOs of the boron moiety. Our results provide a hint for the development of boronate covalent organic frameworks.
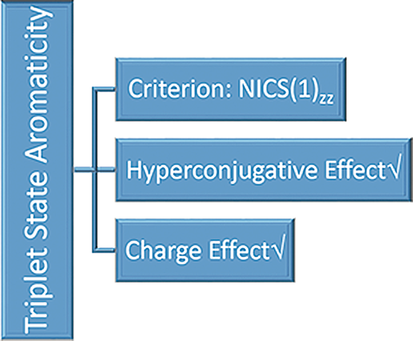
Aromaticity, one of the most important concepts in organic chemistry, has attracted considerable interest from both experimentalists and theoreticians. It remains unclear which NICS index is best to evaluate the triplet-state aromaticity. Here, we carry out thorough density functional theory (DFT) calculations to examine this issue. Our results indicate that among the various computationally available NICS indices, NICS(1)zz is the best for the triplet state.
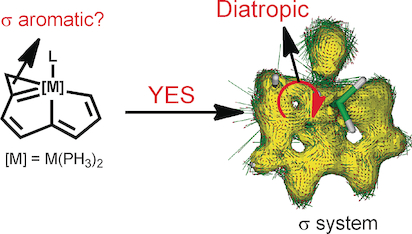
Aromaticity, an old but still fantastic topic, has long attracted considerable interest of chemists. Generally, π aromaticity is described by π-electron delocalization in closed circuits of unsaturated compounds whereas σ-electron delocalization in saturated rings leads to σ aromaticity. Interestingly, our recent study shows that σ aromaticity can be dominating in an unsaturated three-membered ring (3MR) of cyclopropaosmapentalene. An interesting question is raised: Can the σ aromaticity, which is dominant in the unsaturated 3MR, be extended to other cyclopropametallapentalenes?
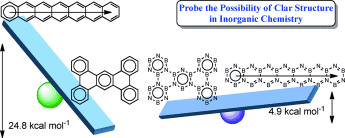
The Clar structure of polybenzenoid hydrocarbons (PBHs) have attracted considerable interest of both theoretical and experimental chemists since it was proposed in the 1950s. However, it remains unclear whether the Clar structure could exist in inorganic PBHs, the boron nitride (BN) analogues where the alternate boron and nitrogen atoms are used to replace the carbon atoms of PBHs. Here, we carry out thorough density functional theory (DFT) calculations to probe the possibility of Clar structures in BN analogues of PBHs.
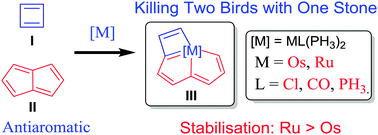
Density functional theory (DFT) calculations were carried out to investigate the stability and aromaticity of metallapentalocyclobutadienes. The results reveal unexpected higher stabilisation achieved with a 3d ruthenium fragment compared to the 4d osmium counterpart. Moreover, direct 1–3 metal–carbon bonding in the metallabutadiene unit of these two complexes is negligible.
http://pubs.rsc.org/en/content/articlelanding/2015/cc/c5cc08291a#!divAbstract
For details, please check the link at http://onlinelibrary.wiley.com/doi/10.1002/asia.201500897/abstract
Communication
Unexpected Higher Stabilisation of Two Classical Antiaromatic Frameworks with a Ruthenium Fragment over Osmium Counterpart: Origin Probed by DFT Calculations
Jingjing Wu, Yulei Hao, Ke An and Jun Zhu
For details, please check the link at http://onlinelibrary.wiley.com/doi/10.1002/cphc.201500811/abstract.
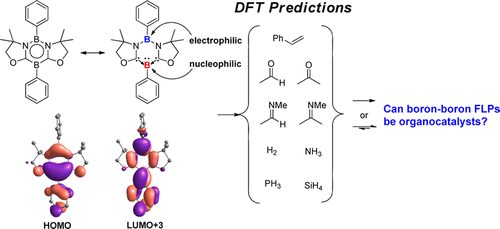
Recent progress in frustrated Lewis pairs (FLPs) has attracted increasing attention. However, most of the FLPs are composed of Lewis basic phosphines and Lewis acidic boranes. In 2015, Kinjo and co-workers reported the first intramolecular boron–boron FLP, namely, 1,3,2,5-diazadiborinine (1), which showed high regioselectivity in the reactions with methyl trifluoromethansulfonate, phenylacetylene, and CO2. More interestingly, the activation of CO2 was found to be reversible when the temperature was elevated to 90 °C.
Copyright © 2025,
Theme Originally Created by Devsaran
