Probing Hyperconjugative Aromaticity of Monosubstituted Cyclopentadienes
Submitted by Jun Zhu on Fri, 01/11/2019 - 20:13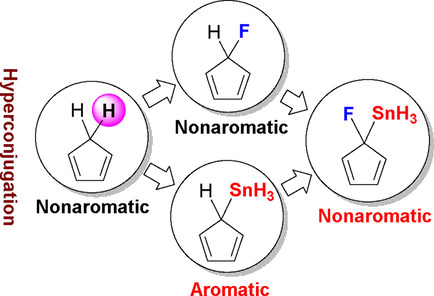
Hyperconjugation and aromaticity are two of the most important concepts in chemistry. Mulliken and co‐workers combined both terms to explain the stability of cyclopentadiene. Here, we carried out DFT calculations on a series of mono‐ and disubstituted cyclopentadiene derivatives to investigate their hyperconjugative aromaticity. Our results revealed that one electropositive substituent can induce aromaticity, whereas one electronegative substituent prompts nonaromaticity rather than antiaromaticity.
