Neutral All-Metal σ-Aromaticity in a Rhombic Cluster
Submitted by Jun Zhu on Mon, 04/21/2025 - 15:12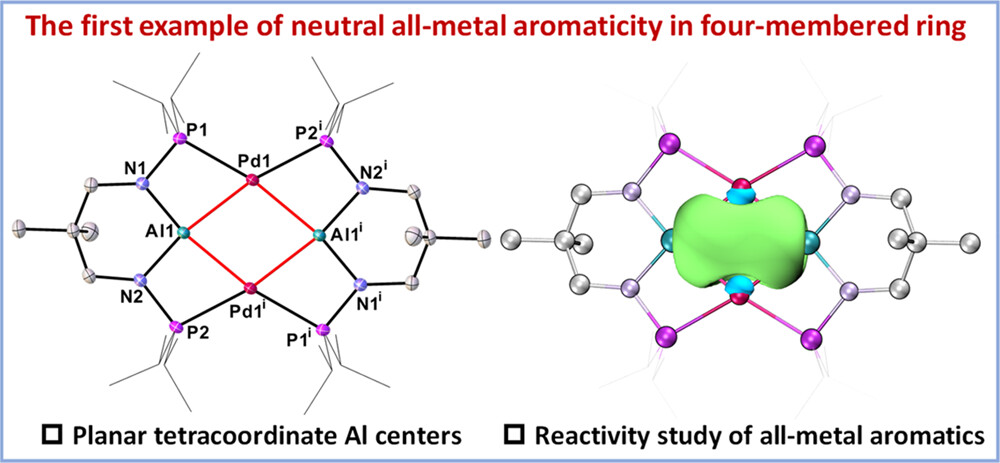
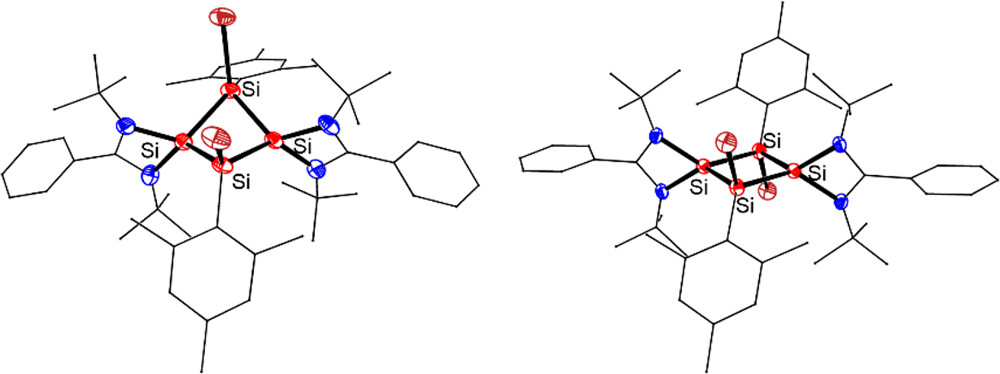
Aromaticity is a cornerstone concept in chemistry, playing a crucial role in understanding molecular stability and reactivity. Traditionally, aromaticity has been primarily associated with cyclic planar conjugated organic molecules composed solely of carbon, but it has recently expanded to include metal-containing systems. However, metal-only aromatics remain extremely scarce. Here, we present the first neutral all-metal aromatic cluster with a rhombic geometry.