Fragmentation of pentacoordinate spirobicyclic aminoacylphosphoranes (P-AAs) by electrospray ionization tandem mass spectrometry concerning P-O and P-N bond cleavage
Submitted by Jun Zhu on Fri, 11/01/2013 - 00:58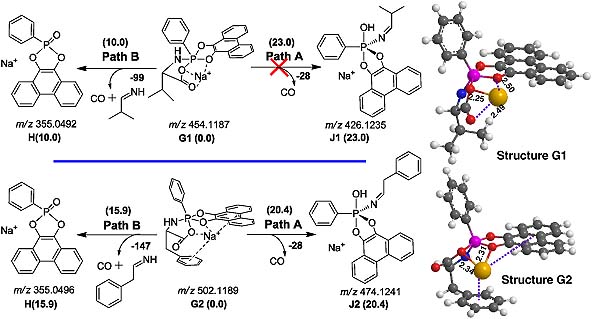
The fragmentation pathways of both protonated and sodiated pentacoordinate spirobicyclic aminoacylphosphoranes (P-AAs) have been studied by electrospray ionization multi-stage mass spectrometry (ESI-MSn) in positive mode. The possible pathways and their mechanisms are elucidated through the combination of ESI-MS/MS, isotope (15 N and 2H) labeling and high-resolution Fourier transform ion cyclotron resonance (FTICR)-MS/MS. The relative Gibbs free energies (ΔG) of the product ions and possible fragmentation pathways are estimated at the B3LYP/6-31 G(d) level of theory.
