A Bis(silylene)pyridine Pincer Ligand Can Stabilize Mononuclear Manganese(0) Complexes: Facile Access to Isolable Analogues of the Elusive d7-Mn(CO)5 Radical
Submitted by Jun Zhu on Thu, 07/07/2022 - 07:45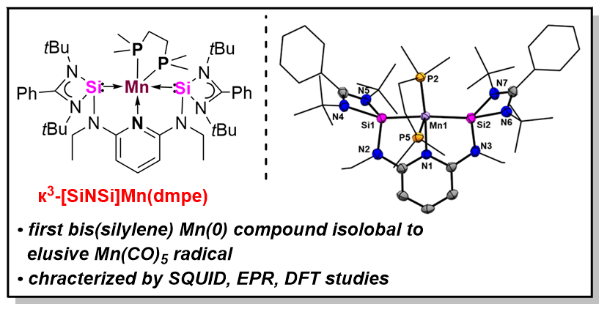
Using the potentially tridentate N,N’-bis(N-heterocyclic silylene)pyridine [SiNSi] pincer-type ligand, 2,6-N,N’-diethyl-bis[N,N’-di-tert-butyl(phenylamidinato)silylene] diaminopyridine, led to the first isolable bis(silylene)pyridine-stabilized manganese(0) complex, {к3-[SiNSi]Mn(dmpe)} 4 (dmpe = (Me2P)2C2H4), which represents an isolobal 17 VE analogue of the elusive Mn(CO)5 radical.