Probing the Limit of the Number of Saturated Atoms for Achieving Hyperconjugative Aromaticity
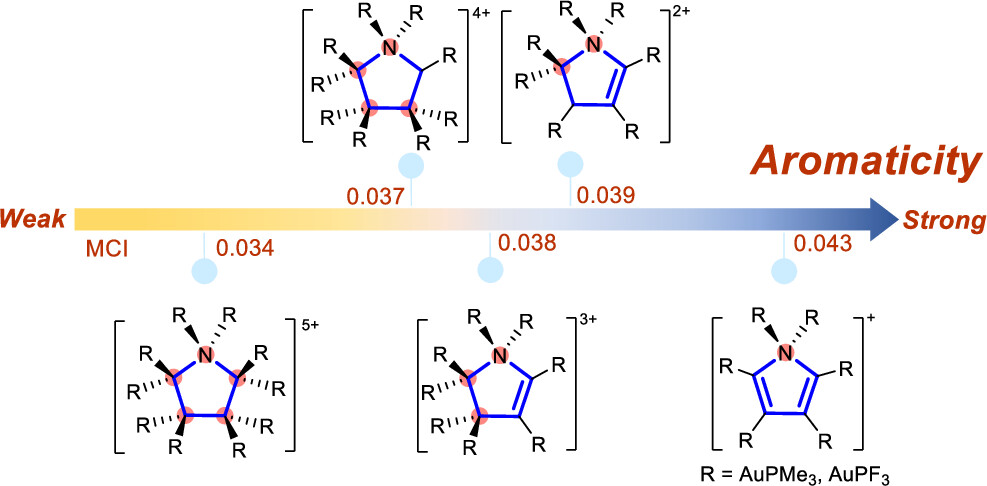
Aromaticity is a fundamental concept in organic chemistry. Hyperconjugative aromaticity, also known as hyperconjugation-induced aromaticity, has evolved from its origin from main group substituents to transition metal analogues, establishing itself as an important category of aromaticity. Additionally, aromatic compounds comprising two sp3-carbon atoms have recently been reported both experimentally and computationally. However, what is the maximum number of sp3-hybridized atoms needed to maintain hyperconjugative aromaticity? Here, we report that hyperconjugative aromaticity can be achieved in hexa-substituted indoliums and octa-substituted pyrroliums, possessing three-five sp3-hybridized carbon/nitrogen atoms by means of density functional theory (DFT) calculations. The aromaticity was confirmed by using various aromaticity indices, i.e., NICS, MCI, and EDDB. Notably, the strong electron-donating ability and aurophilicity of Au(I) substituents play a pivotal role in maintaining the aromaticity and structural integrity. In addition, increasing the number of hyperconjugative centers will decrease the aromaticity in these five-membered rings. Our findings highlight the significance of transition metal substituents in hyperconjugative aromaticity and offer a novel approach for designing aromatic organometallics.
