NO-induced adaptive antiaromaticity in cyclobutadiene
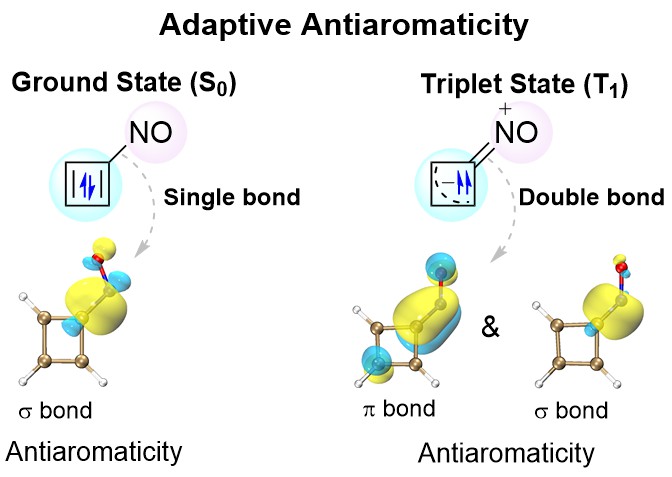
Cyclobutadiene (CBD) displays aromaticity in the lowest-lying triplet excited state (T1) according to Baird's 4n electron rule. Hence, antiaromatic CBD in the T1 state has never been reported so far. Here we demonstrate via density functional theory (DFT) calculations that the CBD ring could possess dual antiaromaticity in the lowest singlet state (S0) and T1 states (termed as adaptive antiaromaticity), which is supported by various aromaticity indices including NICS, ACID, ΔBL, ELF and ISE. Furthermore, the spin density localization on the substituent is the key factor for NO-substituted CBD to maintain antiaromaticity in the T1 state. The principal interacting spin orbital (PISO) analysis suggests that the nitrogen atom in the NO substituent tends to form a double bond with the carbon atom in the CBD ring, blocking the delocalization into the CBD ring and resulting in the antiaromaticity in the T1 state. Our findings represent a step forward in aromatic chemistry.
https://pubs.rsc.org/en/content/articlelanding/2023/QO/D3QO01498C
