Requirement for an oxidant in Pd/Cu co-catalyzed terminal alkyne homocoupling give symmetrical 1,4-disubstituted 1,3-diynes
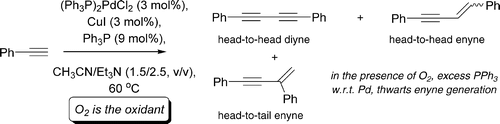
Palladium-catalyzed terminal alkyne dimerization, through oxidative homocoupling, is a useful approach to the synthesis of symmetrical 1,4-diynes. Recent investigations have suggested that this reaction might be accomplished in the absence of intentionally added stoichiometric oxidants (to reoxidize Pd(0) to Pd(II)). In this paper, we have fully addressed the question of whether oxygen (or added oxidant) is required to facilitate this process. The presence of a stoichiometric quantity of air (or added oxidant such as I2) is essential for alkyne dimerization. Excess PPh3 inhibits alkyne dimerization to enyne, which only occurs to a significant extent when the reaction is starved of oxidant. Theoretical studies shed more light on the requirement for an oxidant in the homocoupling reaction in order for the process to be theromodynamically favorable. The employment of I2 as the stoichiometric oxidant appears to be the method of choice. The dual role of Cu both in transmetalation of alkynyl units to Pd(II) and in assisting reoxidation of Pd(0) to Pd(II) is suggested.
