Synthesis of Aromatic Aza-metallapentalenes from Metallabenzene via Sequential Ring Contraction/Annulation
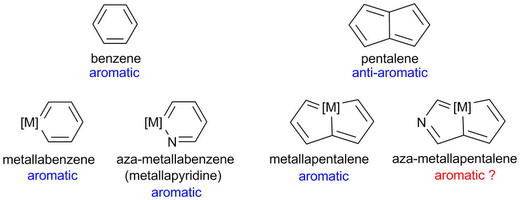
The concept of aromaticity has long played an important role in chemistry and continues to fascinate both experimentalists and theoreticians. Among the archetypal aromatic compounds, heteroaromatics are particularly attractive. Recently, substitution of a transition-metal fragment for a carbon atom in the anti-aromatic hydrocarbon pentalene has led to the new heteroaromatic osmapentalenes. However, construction of the aza-homolog of osmapentalenes cannot be accomplished by a similar synthetic manipulation. Here, we report the synthesis of aza-osmapentalenes by sequential ring contraction/annulation reactions of osmabenzenes via osmapentafulvenes. Nuclear magnetic resonance spectra, X-ray crystallographic analysis, and DFT calculations all suggest that these aza-osmapentalenes exhibit aromatic character. Thus, the stepwise transformation of metallabenzenes to metallapentafulvenes and then aza-metallapentalenes provides an efficient and facile synthetic route to these bicyclic heteroaromatics.
http://www.nature.com/srep/2015/150402/srep09584/full/srep09584.html
